Top EDC software solutions for clinical trial teams
Clinical trials are often delayed due to operational inefficiencies, particularly when data and workflows are spread across disconnected systems. In such environments, even well-designed studies can be affected by data inconsistencies, increased error rates, and rising operational costs.
This is where choosing the right electronic data capture (EDC) system becomes critical. Traditional EDC solutions were built as standalone tools, useful for data entry, but not for the complex, interconnected workflows modern trials demand. As a result, teams face issues like duplicated effort, manual reconciliation, and slower decision-making.
A unified EDC system changes that. By centralizing study builds, data management, and operational oversight, it streamlines processes, improves accuracy, and reduces compliance risks. But with so many vendors offering similar-sounding features, it can still be challenging to identify which solution truly supports end‑to‑end trial execution.
In this blog, we’ll walk through the top EDC software solutions and help you understand which one is the best match for your clinical trial.
What to look for in an EDC system
In Wemedoo’s “Clinical trial trends in 2025” overview, unified systems and connected infrastructures are highlighted as essential drivers of efficiency in modern clinical research. That’s why, when evaluating EDC software platforms, clinical teams should look beyond individual features and assess how well the system supports the full trial workflow, data flow, and long‑term scalability. In a truly unified system, all trial functions operate natively in one environment, eliminating integrations, manual corrections, and copy‑pasting, resulting in minimal data cleaning and no hidden integration costs or delays.
Key factors to consider:
1. Workflow efficiency
- Tools that streamline daily tasks and reduce manual effort
- Configurable, intuitive study‑build capabilities
- Real-time visibility into site and patient progress to prevent operational blind spots
2. Minimizing operational friction
- Avoid systems that require frequent manual reconciliation
- Watch for outdated interfaces or tools that operate in isolation
- Prioritize solutions that prevent added work rather than create it
3. Unified and interoperable ecosystem
- Ability for data to move seamlessly across modules
- Support for integrated, end‑to‑end trial processes
- Reduction of fragmented systems that slow teams down
4. Regulatory and quality readiness
- Comprehensive audit trails
- Validated workflows that withstand inspection
- Native alignment with global compliance frameworks
- Support for inspection‑ready execution from start to finish
5. Secure access and identity management
- Single Sign‑On (SSO) as a native component, enabling centralized identity management and role‑based access control across all system modules
- Simplified system use with lower risk of access errors
- Continuous compliance with SOPs, industry standards, and regulatory requirements, even as they evolve over time
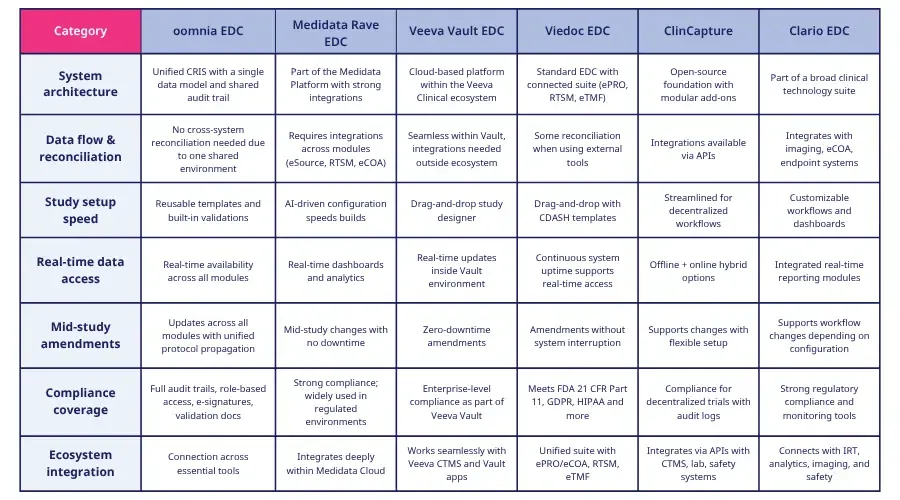
oomnia EDC software
oomnia EDC is an electronic data capture system built within a unified clinical research information system (CRIS), designed to allow teams to manage study data in a single environment and reduce reliance on separate tools. It’s built to provide consistent data flow, real‑time access to study information, and streamlined setup activities within the broader platform.
Well-suited for: Small and mid‑sized CROs, growing biotech, medtech, academia, and anyone who needs a complex study setup with innovative randomization schema and native full CDISC standards support.
Key advantages:
- Unified environment: Built as a part of a single, semantically interoperable CRIS, designed to reduce tool fragmentation and duplicated data entry.
- Real‑time data access: Study data can be available for monitoring and review in real-time.
- Streamlined setup and change management: Configurable electronic case report forms (eCRFs) and reusable templates help standardize study builds and allow protocol updates to be deployed without interrupting site workflows.
- Standardized structures: Shared terminology, IDs, and data models across studies.
- Compliance support: Built to include audit trails, access controls, electronic signatures, and validation documentation.
- Global deployment: Worldwide study execution supported with continuous 24/7 support
- Native advanced insights and reporting: Built to provide interactive views and configurable reports, offering deeper visibility into study progress, data quality, and operational performance.
Medidata Rave EDC
Medidata Rave EDC is an electronic EDC software designed to offer teams a system for capturing, managing, and reporting trial data across all phases and therapeutic areas.
Well-suited for: Large global pharma companies and mid‑to‑large CROs running complex, multinational Phase I–IV trials requiring scale, standardization, and broad ecosystem integrations
Key advantages:
- Proven industry adoption: Used in over 38,000 trials.2
- AI‑enhanced study design: It can automate configuration tasks to speed study builds and reduce setup time.
- Advanced analytics and reporting: Real‑time dashboards and robust data review tools support faster decision‑making and operational oversight.
- Strong interoperability: Facilitates centralized data handling and integration with the broader Medidata Clinical Cloud.
- Flexible study support: Capable of handling a wide range of study sizes and protocol complexities across therapeutic areas.
Veeva Vault EDC
Veeva Vault EDC is a cloud‑based electronic data capture system designed to support clinical data collection within the Veeva platform. It’s built to provide standard EDC functionality and support protocol amendments through configurable study designs, with integration into other Veeva clinical systems.
Well-suited for: Mid‑sized to large sponsors and CROs already using the Veeva ecosystem who need an EDC solution that can accommodate protocol amendments and alignment with related clinical operations tools.
Key advantages:
- Zero‑downtime amendments: Protocol changes can be deployed without interrupting site workflows.
- Modern cloud infrastructure: Built for speed, scalability, and reliable performance across global trials.
- Intuitive design tools: Includes a drag‑and‑drop study designer for faster configuration.
- Dynamic study execution: Supports dynamic visits and forms for more flexible protocol designs.
- End‑to‑end environment: Comprehensive environment designed for collecting and processing site‑reported patient data across all phases.
Viedoc EDC
Viedoc EDC is an electronic data capture system designed to support clinical trials. It provides tools for collecting and managing site‑reported data, offers standard compliance features, and includes workflows shaped by CRO experience.
Well-suited for: Small to mid‑sized sponsors, CROs, and device companies needing fast setup, intuitive workflows, and strong global compliance support.
Key advantages:
- Faster study builds: Drag‑and‑drop interface and CDASH‑ready templates accelerate startup timelines.
- High system reliability: 99% uptime ensures continuous availability during active studies.3
- Mid‑study flexibility: Built to make amendments without system interruptions.
- Global regulatory compliance: Meets FDA 21 CFR Part 11, GDPR, HIPAA, and regional requirements in Japan and China.
- Global reach: Supports 40+ languages and offers 24/7 technical assistance across multiple international offices.3
ClinCapture Captivate EDC
ClinCapture offers an EDC software is built around the Captivate platform, providing tools for collecting and managing trial data in decentralized and hybrid study settings. It is designed to support various levels of study complexity and includes capabilities for offline data capture and integration with other clinical systems.
Well-suited for: Small biotech, academic teams, and mid‑sized CROs seeking a budget‑flexible, customizable, private‑cloud EDC suited for decentralized and hybrid trials.
Key advantages:
- Decentralized trial focus: Designed to support remote and hybrid data collection workflows
- Open‑source foundation: Enables flexible development and customization where needed
- Cross‑study reporting: Supports reporting across multiple sites and studies within the platform
- System integrations: Offers APIs and web services for CTMS, lab, and safety system connectivity
- Private cloud option: Controlled deployment in dedicated cloud environments
Clario EDC
Clario provides EDC software as part of a broader suite of clinical technologies, offering tools for data capture alongside solutions for imaging, eCOA, cardiac safety, and endpoint management. It is designed to support regulated studies with integrated workflows and continuous customer support.
Well-suited for: Large global sponsors and mid‑sized biotech running complex, multi‑arm trials that require integrated endpoint technologies and strong site usability.
Key advantages:
- Established global provider: Long-standing industry experience across diverse clinical trial solutions
- Strong regulatory compliance: Built‑in audit trails and compliance monitoring to support regulated studies
- Integrated clinical solutions: Connects with imaging, eCOA, and endpoint data capture tools
- 24/7 support: Provides continuous customer and patient assistance
- Customizable workflows: Includes configurable dashboards and site workflows
- System interoperability: Integrates with interactive response technology (IRT), analytics, and reporting modules for streamlined processes
EDC softwares overview
Choosing the right EDC software to move your trials forward
This article reflects our perspective on the EDC market and highlights solutions based on publicly available information and our experience as a platform provider.
Selecting the right EDC software is ultimately about empowering clinical teams with tools that reduce complexity, improve data quality, and speed up trial execution. While each software covered in this guide offers meaningful capabilities, their differences become clear when looking at how they handle data flow, system interoperability, compliance readiness, and the day‑to‑day struggles of running modern studies.
Unified EDC environments can reduce the operational friction that often occurs when teams manage data across multiple disconnected systems. In contrast, fragmented or tool‑heavy setups typically introduce extra reconciliation steps, duplicate work, and slower access to study insights.
When selecting an EDC system or evaluating different EDC platforms, the choice often depends on the level of integration and operational complexity your study requires. Unified softwares are generally a good fit for teams that want all core trial functions in one environment, minimizing the need for data reconciliation between systems. Ecosystem‑based tools work well for organizations that prefer modular flexibility and want to integrate best‑in‑class solutions across different vendors. Traditional standalone EDC systems may be more suitable for studies with narrower requirements or teams that only need data capture without broader operational components.
Now that you’ve explored the top EDC software solutions, the next step is choosing the software that best aligns with your workflow and long‑term clinical research strategy. See whether oomnia’s unified architecture offers the structure your studies require and explore how oomnia EDC software can support your clinical trial execution.
This article is for informational purposes only and reflects our perspective based on publicly available information at the time of writing. Product names and trademarks are the property of their respective owners and are used for identification purposes only. Features and capabilities may vary depending on configuration and use.
References
Wemedoo. (2026, January 6). Clinical trial trends in 2025. Accessed March 12, 2026. https://wemedoo.com/blogs/clinical-trials-trends-2025/
https://www.medidata.com/en/clinical-trial-products/clinical-data-management/edc-systems/
https://www.viedoc.com/
https://www.veeva.com/products/veeva-edc/
https://www.captivate.org/captivate-edc/
https://clario.com/

April 14, 2026
Solving MedTech trial complexity with the right technology partner
Blogs

April 2, 2026
What makes a clinical trial software truly unified
Blogs

March 27, 2026
The system architecture behind the data reconciliation challenge in clinical trials
Blogs