The system architecture behind the data reconciliation challenge in clinical trials
Modern clinical trials generate more data than ever, drawing information from a wide range of sources, each with its own format, structure, and quality considerations. As a result, data reconciliation in clinical trials has become one of the most time-consuming operational tasks for clinical data management teams.
For clinical teams, this growing data volume often collides with fragmented technology environments. Instead of supporting efficiency, these disconnected systems slow down data oversight and increase the risk of inconsistencies across the study lifecycle.
As a result, teams regularly find themselves battling operational bottlenecks:
- Reconciling mismatched datasets
- Investigating the source of discrepancies
- Managing endless email chains between vendors and internal teams
- Switching between multiple spreadsheets just to stay on top of data oversight
In practice, data reconciliation problems in clinical trials are rarely caused by the data itself, but the way clinical trial systems are structured and connected. These manual processes not only delay trial execution but also increase the risk of errors that can impact timelines, budgets, and ultimately regulatory submissions.
As clinical research management grows in scale and complexity, many organisations are turning toward unified, system‑based approaches to bring structure and transparency back to the clinical data lifecycle.
This article builds on our previous discussions about the limitations of modular eClinical architectures and the compliance risks they introduce in modern clinical trials, exploring how these structural challenges directly affect one of the most resource-intensive activities in study execution: data reconciliation.
Data fragmentation across clinical trial systems
One of the main drivers of reconciliation complexity is data fragmentation across systems.
Trial data is typically distributed across multiple environments including Electronic Data Capture (EDC) systems, laboratory databases, safety systems, imaging repositories, and vendor-specific platforms. Each system operates with its own processes, formats, and data rules.
Without a unified setup or shared data structures, information must often be transferred manually between systems. This introduces additional effort and creates opportunities for inconsistencies. This structural fragmentation is closely related to the architectural limitations of modular eClinical architectures. As discussed in our article on “Why eClinical architectures have reached their limits,” many clinical technology ecosystems evolved by connecting independent tools rather than designing a unified data foundation from the beginning.
For years, multiple resources reported that fragmented and incompatible data slows analysis and delays real‑world impact, demonstrating why unified and connected data platforms are necessary in clinical trials.¹,²,³
Within a single study, data managers, statisticians, clinical operations, and external partners may all work in the same datasets, yet lack a shared, real‑time view of the study
As a result:
- Issues can be identified late
- Discrepancies may be duplicated across teams
- Data problems may be resolved inconsistently
In response to these challenges, clinical teams often try to fix gaps by adding yet another tool to an already crowded technology landscape. While this can temporarily address specific problems, it usually creates new silos, adds extra reconciliation steps, and increases the overall complexity of day‑to‑day work.
Instead of stacking more solutions on top of existing problems, the real goal should be a unified platform that brings data, workflows, and oversight together in one place, reducing duplication and giving every stakeholder a clear, shared view of study progress.
The impact of diverse clinical data on fragmentation
Modern clinical trial phases rely on a wide range of data sources, each generated through different technologies and systems.
Key data types that increase fragmentation include:
- EDC and clinical assessments
- Laboratory and biomarker data
- Imaging data
- Device‑generated and sensor data
- Patient‑reported outcomes (ePRO/eDiary)
- Safety and pharmacovigilance data
Individually, each dataset adds valuable clinical insight. However, when these sources remain disconnected, they create an environment where mismatches, gaps, and inconsistencies can quickly appear.
As trials expand and incorporate more tools and external vendors, fragmentation intensifies, making reconciliation an increasingly complex and manual task.
These challenges also highlight the growing importance of clinical data interoperability, which we explored further in our article on interoperability as the missing piece in clinical trial efficiency.
How does data fragmentation affect clinical trial execution?
When data is distributed across multiple systems, it becomes difficult for study teams to maintain a timely and complete view of trial progress. Important updates may be delayed or overlooked, which can affect patient safety monitoring and operational decision-making.
Fragmentation also slows trial execution because teams must spend extra time comparing datasets, checking versions, and resolving discrepancies rather than focusing on study oversight. From a compliance point of view, scattered data makes it difficult to meet regulatory expectations and follow Attributable, Legible, Contemporaneous, Original, and Accurate (ALCOA+) principles, which require data to be accurate, complete, consistent, and easy to trace.
As detailed in our article “Why Compliance Risk Increases in Modular eClinical Setups,” fragmented systems, particularly those built on modular eClinical architectures, can introduce broader compliance risks when audit trails and data ownership are spread across multiple tools.
When information is missing or stored in different formats, it becomes harder to demonstrate that the trial data is complete, consistent, and traceable, increasing the risk of inspection findings. These bottlenecks are well‑documented, and reliance on spreadsheet‑based oversight further heightens inspection risk due to poor traceability and limited auditability.
The solution is straightforward: unified systems like oomnia keep all data in one place, making it easier to spot risks early and maintain a clean, audit‑ready record throughout the study.
What is data reconciliation in clinical trials?
Data reconciliation in clinical trials refers to the process of ensuring that external datasets required by the study match the corresponding information in the EDC system.
This typically involves comparing key identifiers such as:
- Subject numbers
- Visits and time points
- Collection dates
- Laboratory results or safety information
Any mismatch, missing entry, conflicting value, or structural inconsistency must be resolved by the appropriate party, whether that’s the external vendor, site staff, or clinical operations team.
To guide this effort, sponsors, Contract Research Organisations (CROs), and vendors typically define reconciliation requirements in advance through a data cleaning or reconciliation plan.
Because reconciliation requires visibility across multiple data sources, unified clinical trial software oomnia can make reconciliation more straightforward by allowing teams to review and resolve issues in one environment rather than navigating several disconnected systems. ⁴
This unified approach reduces manual effort and supports a more consistent, reliable data foundation for downstream analysis and regulatory submission.
Why clinical trial system architecture matters
The underlying architecture of clinical trial software has a direct impact on how efficiently reconciliation can be performed. In many organisations, this architecture takes the form of modular eClinical systems where individual trial functions operate as separate tools connected through integrations.
In fragmented setups, study data resides across separate tools that only partially connect. Teams must manually compare datasets, transfer files, and investigate discrepancies across systems.
In other words, data reconciliation complexity is often an architectural issue rather than simply a data management task. This slows down data cleaning, increases the chance of missed updates, and makes it harder to maintain a complete, trustworthy audit trail. At the same time, reconciliation challenges can arise from factors beyond architecture, such as poor site documentation or vendor‑related errors, which makes a strong system foundation even more critical.
Unified architecture works differently. Instead of distributing trial data across separate tools, all core study activities, data capture, review, reconciliation, and oversight, live in one coordinated environment. This reduces system handoffs and eliminates many gaps where errors typically occur, while still relying on strong governance and clear processes to ensure the system operates effectively.
Unified clinical research information systems (CRIS) like oomnia follow this approach by bringing essential trial functions together in a single clinical trial system. This helps teams avoid the constant back‑and‑forth between disconnected tools and supports a cleaner, more transparent data lifecycle.
Modular eClinical systems vs. unified clinical trial software
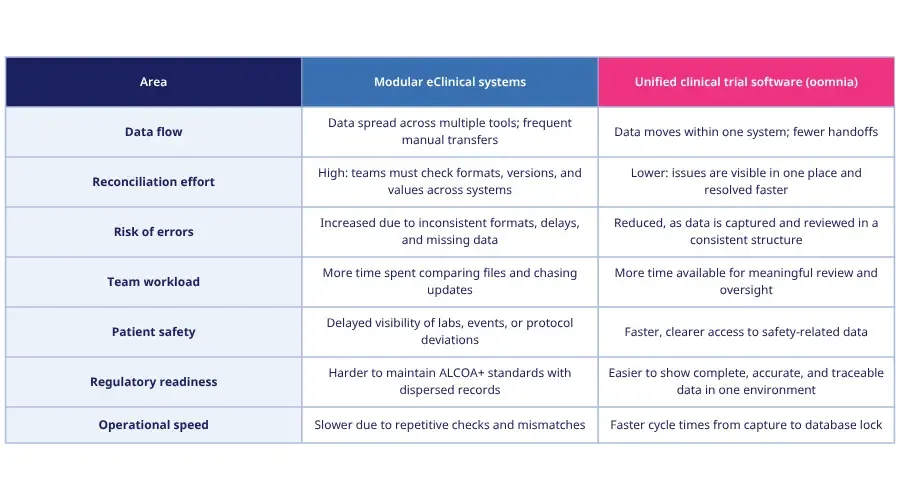
How oomnia helps simplify data reconciliation
The challenges created by fragmented systems, manual data handling, and inconsistent workflows highlight the need for a more connected clinical trial software.
oomnia addresses these challenges through a unified, interoperable architecture that brings all essential trial tools into one coordinated environment.4 Instead of relying on separate systems for EDC, ePRO, eTMF, eConsent, eSource, RTSM, and CTMS, oomnia aligns these tools within one system where data flows consistently from capture to submission.
Because data and workflows operate within the same environment:
- Teams do not need to switch between multiple systems
- Data does not need to be manually transferred between tools
- Discrepancies can be identified and resolved earlier
For data managers, monitors, and study leads, this means clearer oversight, faster issue resolution, and fewer operational delays.
Through the centralization of tools and creation of real interoperability, oomnia supports a cleaner, more reliable data lifecycle, which strengthens patient‑safety monitoring, improves regulatory readiness, and gives teams more time to focus on meaningful work rather than administrative tasks.
Reducing data reconciliation effort in clinical trials increasingly depends on system architecture rather than manual processes alone.
In an environment where trial complexity continues to grow, a unified clinical research information system (CRIS) like oomnia provides the support needed for efficient, consistent, and high‑quality clinical trial execution.
Ready to see how a unified system can improve your trial?
Learn more about oomnia and explore how it can simplify workflows for your clinical team.
References
Bhatt, J., Gaikwad, A., Garudasu, S., Goel, O., Jain, A., & Singh, N. (2024). Addressing data fragmentation in life sciences: Developing unified portals for real-time data analysis and reporting. IRE Journals, 8(4), 641. https://www.irejournals.com/formatedpaper/1706397.pdf
Building a scientific narrative towards a more resilient EU society . Part 1, A conceptual framework, Publications Office, 2017, https://data.europa.eu/doi/10.2760/635528
Wei, W. Q., Leibson, C. L., Ransom, J. E., Kho, A. N., Caraballo, P. J., Chai, H. S., Yawn, B. P., Pacheco, J. A., & Chute, C. G. (2012). Impact of data fragmentation across healthcare centers on the accuracy of a high-throughput clinical phenotyping algorithm for specifying subjects with type 2 diabetes mellitus. Journal of the American Medical Informatics Association : JAMIA, 19(2), 219–224. https://doi.org/10.1136/amiajnl-2011-000597
Wemedoo. (2024). Interoperability: The missing piece in clinical trial efficiency. Accessed March 3, 2026. https://wemedoo.com/blogs/interoperability-the-missing-piece-in-clinical-trial-efficiency-1/

April 28, 2026
Top EDC software solutions for clinical trial teams
Blogs

April 14, 2026
Solving MedTech trial complexity with the right technology partner
Blogs

April 2, 2026
What makes a clinical trial software truly unified
Blogs