What makes a clinical trial software truly unified
In clinical research, many of the daily frustrations teams experience all trace back to the same underlying cause: fragmented system architecture. Most clinical trial solutions are built as modular eClinical architectures, where separate systems operate independently rather than as one unified environment, leaving teams to manage a patchwork of tools that never truly work together.
These problems are not operational inconveniences; they are architectural consequences. When each module maintains its own data structures, workflows, and logic, teams inevitably face compliance risk, extra oversight, and continuous reconciliation just to keep a trial running smoothly.
So, the real question becomes: What would clinical trial software look like if these issues were solved at the architectural level instead of managed through integrations? The answer is a truly unified software built as a clinical research information system (CRIS), where data, processes, and stakeholders operate in one native environment.
This article explores what defines such a unified system, how it differs from the modular eClinical architectures the industry has long relied on, and why solving fragmentation at the architectural level changes what’s possible in clinical trials.
Where fragmentation creates risk, rework, and delays
As highlighted in Wemedoo’s analysis of modular eClinical system limitations “Why eClinical architectures have reached their limits, and a new category is emerging”, today’s research has become too complex for the old system designs to keep up, leading to ongoing problems that small adjustments can’t solve.
Fragmentation forces teams to work around their tools instead of through them, turning simple tasks into slow handoffs, duplicate entry, and constant reconciliation just to keep information consistent. That slowdown appears in inspection prep, audit trails, and daily oversight, issues outlined in our blog “Why compliance risk increases in modular eClinical setups,” which explains how integration gaps lead to duplicated validation work, disconnected audit trails, and reactive, cross‑vendor coordination.
In parallel, our article “The system architecture behind the data reconciliation challenge in clinical trials” explains how scattered data across EDC, CTMS, ePRO, labs, and devices turns reconciliation into a permanent workload, slowing database lock, delaying decisions, and increasing the risk of inconsistencies that must be chased across systems.
These issues all come from the same place: the system’s architecture. They aren’t random operational problems, but the natural result of running trials on a modular, fragmented setup. These problems will not disappear until the software is unified from the ground up as a CRIS.
Why integration is not the same as unification
Integration and unification are often mistaken for the same thing, but they address completely different challenges. Integrating systems only connect separate tools so they can exchange data, while each system continues to operate with its own workflows, logic, and data structures.
Unification goes deeper: it means there is only one system, so nothing needs to be connected or reconciled in the first place.
Integrating systems can make workflows seem smoother, but it never removes the underlying complexity. Every integration still carries the risk of mismatched data, duplicated configuration, and additional oversight. Even when integrations work well, teams remain responsible for managing multiple audit trails, validation packages, and process handoffs across different tools.
A clinical research information system (CRIS) approach represents a fundamentally different architectural model. Instead of stitching together modules, a CRIS is built as a single environment where data, workflows, and stakeholders operate natively together.
This shift from “connected systems” to “one system” is what makes true unification possible and removes the structural friction that slows trials down. It’s a distinction that Valérie Czaya, Wemedoo’s Chief Operating Officer, summarizes clearly: “In most clinical trial environments, the problem isn’t how systems connect, it’s that they were never designed to operate as one system in the first place.”
What defines a unified clinical trial software
A unified clinical trial software is built on a single architectural foundation, not a collection of tools stitched together. When software is built as a clinical research information system (CRIS), it follows a set of essential principles that make true unification possible.
A unified system starts with a shared clinical data foundation, meaning all study information, operational, clinical, and patient‑reported, is created and stored within the same environment. Because everything lives in one place, the system operates on a single data model rather than multiple disconnected structures that require mapping or translation.
This foundation also enables native workflows, allowing every activity to happen inside the same system including:
- Data capture
- Monitoring
- Documentation and operational oversight
Finally, a unified software provides a single audit trail that captures every action across the entire trial. With all data and workflows inside one environment, oversight becomes simpler, cleaner, and compliant, without the need to combine logs from different tools.
Together, these principles define what “unified” truly means: not integrated systems, but one system built to support the full lifecycle of clinical trial.
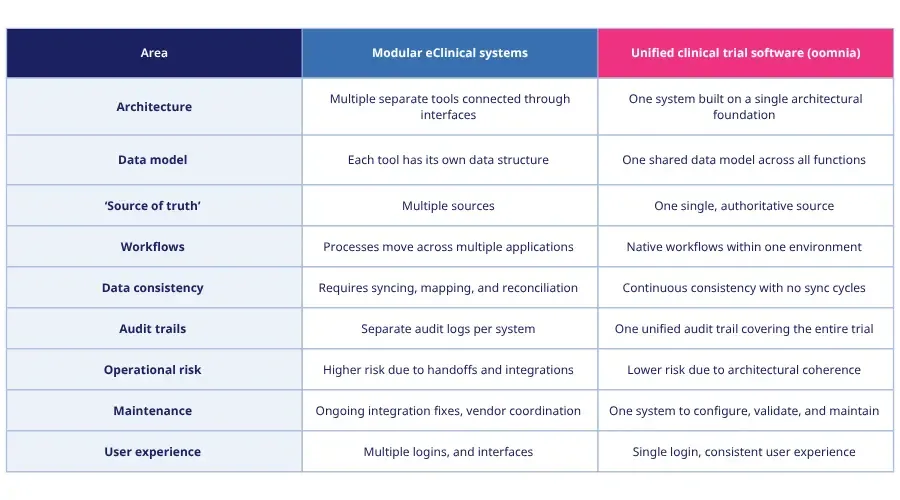
Integrated tools or one unified system? A quick comparison
A simple way to understand the difference between integrated and unified systems is to look at how each handles data, workflows, and oversight. The table below highlights the key differences.
Choosing clinical trial software built on true unification
When we sum everything up, choosing the right clinical trial software becomes an architectural decision, not just a feature comparison. Many solutions today present themselves as “unified,” but the real question is: what is that unification built on? Is it created through integrations that connect separate modules, or is it built as a single system from the ground up?
A truly unified solution follows CRIS principles:
- One shared data foundation
- One unified data model
- Native workflows within a single system
- Continuous consistency
- A unified audit trail across the entire trial
Only systems built this way can avoid the fragmentation and reconciliation burden seen in modular eClinical setups.
oomnia is an example of clinical trial software built on these clinical research information system (CRIS) principles. Rather than linking multiple tools, it operates as one cohesive environment where data and processes naturally work together. Its relevance here is not as a product, but as a demonstration of what a CRIS‑based approach looks like in practice: a system where unification is architectural, not constructed through interfaces.
This distinction matters. When unification is built into the architecture, teams work within one environment designed to support the entire lifecycle of a trial and not a collection of tools that must constantly be held together.
Solving the fragmentation problem at the architectural level
When we look at the challenges clinical trial teams face, constant data reconciliation, rising compliance risk, and persistent workflow fragmentation, it becomes clear that these issues are not isolated incidents. As long as trials run on modular systems stitched together through integrations, these problems will continue to surface, no matter how many processes or tools are added on top.
A CRIS‑based unified software addresses these issues at the root. By operating on one shared data foundation, one data model, native workflows, continuous consistency, and a single audit trail, it removes the structural gaps that create operational friction in the first place. Instead of managing complexity, teams can finally work within a system designed to eliminate it.
If removing reconciliation cycles, reducing compliance risk, and simplifying oversight are priorities for your team, discover how oomnia, a CRIS‑based clinical trial software is built to support trials without the friction of modular setups.
References
Wemedoo. (2026). Why eClinical architectures have reached their limits, and a new category is emerging. Accessed March 20, 2026. https://wemedoo.com/blogs/the-limits-of-eClinical-architectures-and-the-rise-of-CRIS/
Wemedoo. (2026). Why compliance risk increases in modular eClinical setups. Accessed March 20, 2026. https://wemedoo.com/blogs/why-compliance-risk-increases-in-modular-eClinical-setups/
Wemedoo. (2026). The system architecture behind the data reconciliation challenge. Accessed March 20, 2026. https://wemedoo.com/blogs/The-system-architecture-behind-the-data-reconciliation-challenge/

April 28, 2026
Top EDC software solutions for clinical trial teams
Blogs

April 14, 2026
Solving MedTech trial complexity with the right technology partner
Blogs

March 27, 2026
The system architecture behind the data reconciliation challenge in clinical trials
Blogs