With oomnia ePRO patients can report their data anytime via smartphone, tablet, or web. You get cleaner data, higher engagement, and real-time insights that improve both trial quality and patient experience.

Collect Patient Feedback in Real Time & Turn It Into Analysis-Ready Data.


Deliver ePRO questionnaires in any format, length, or language. Easily tailor flows and dynamic responses to meet your study’s unique needs.
Access real-time, integrated graphical reports that combine eCRF and ePRO data for a comprehensive, on-the-spot trial overview.
Monitor data quality and patient compliance as it happens, enabling quicker, smarter decision-making with integrated real-time tracking.
Eliminate transcription errors with direct participant input and automated data aggregation for unmatched accuracy.
Enjoy a programming-free instrument setup with automatic ePRO delivery triggers based on eCRF completion. Accelerate your trial launch.
Reduce manual handling with automated data processing and built-in BYOD support, dramatically lowering your operational expenses.

Inspired by the Latin Word omnia, Meaning 'All Things',
We Created oomnia.
A single, unified system that brings together every part of a clinical trial, from planning to global compliance.
EDC, RTSM, eTMF and CTMS, eSource, ePRO, eCOA and eConsent are integrative parts of one and the same ecosystem supporting multiple trials on a single instance.
Choose the modules and features that work for you and customize everything to fit your workflow.

Protect sensitive patient information with data encryption, multi-factor authentication, and strict role-based access controls.
SAVE UP TO 75% OF NAVIGATION TIME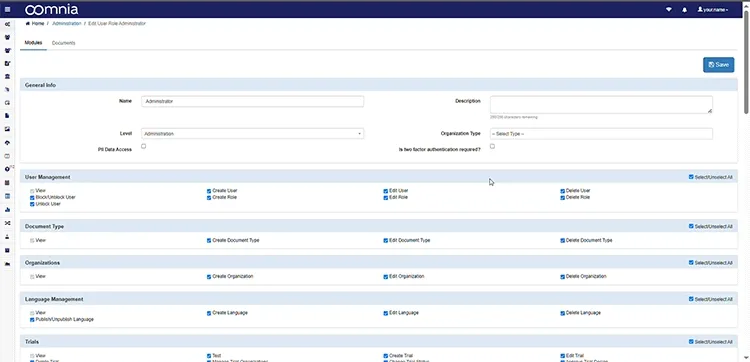
Tailor each interaction with an intuitive interface that adapts to individual needs, driving richer, more insightful responses.
0%PROGRAMMING REQUIRED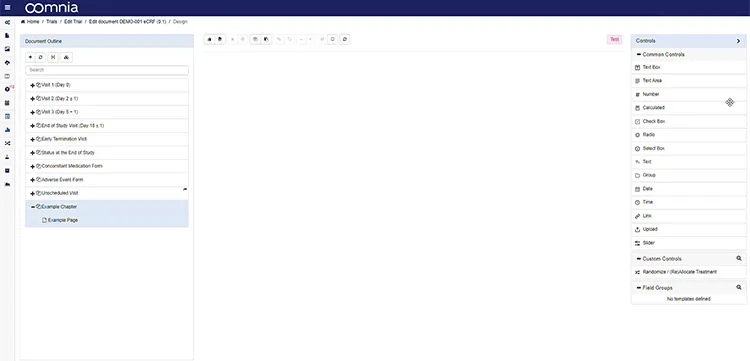
Send notifications to patients to complete their assessments or questionnaires, promoting timely data submission.
REDUCE UP TO 66%OF MANUAL QUERIESoomnia - Meeting Industry Standards
Our unified clinical trial software is designed with compliance at its core, meeting critical industry standards. Our commitment to these regulations ensures that your clinical trials are not only efficient but also adhere to the highest levels of data integrity, privacy, and ethical conduct.




Trusted Partners






















Read more about the experiences from those who have partnered with us

All jobs, both large and small, are performed with the same dedication to quality, detail, innovative thinking, and rigor. It is always a pleasure to work with the Wemedoo team.
Georg Mathis, PhD
CEO Appletree CI Group AG

Wemedoo embodies all the traits desired in a partner in the clinical trial industry – expertise, innovation, reliability, compliance, and attention to detail.
Martina Diminic Smetisko, DMD
Managing Director at Marti Farm Ltd

Their dedication to, and support of, our projects has resulted in clear quality deliverables. They are always there to answer any questions with expertise in a timely manner. I am...
Their dedication to, and support of, our projects has resulted in clear quality deliverables. They are always there to answer any questions with expertise in a timely manner. I am looking forward to our next project together.
Volodymyr Stus, MD
Biopharmaceutics and Clinical Research Department Manager at Polpharma SA

Wemedoo's cross-functional, integrated teams capture EDC set-up, go-live, data capture, data cleaning, and finally data analysis in a globally compliant fashion supporting trials in different geographies including the US. Their...
Wemedoo's cross-functional, integrated teams capture EDC set-up, go-live, data capture, data cleaning, and finally data analysis in a globally compliant fashion supporting trials in different geographies including the US. Their speed of addressing customer needs sets in my personal experience industry benchmark whilst keeping these data solutions lean and affordable. Besides the great proprietary systems they use, their integrated team approach with personal responsibility and honest commitment to targeting outcomes differentiates the Wemedoo team from other providers. Wemedoo only made Oxular's key initial trial, a device-drug trial possible, as based on some pre-work, the Wemedoo team could substantially adapt and update the first EDC ideas for a trial under a US FDA IND. The Wemedoo team worked extremely focused on familiarizing US trial sites and clinical CROs with the use of their system, which turned out to be a key success factor of the respective trial. The highly professional and up-to-date capabilities in data sciences were mirrored by customer-centricity and super fast timelines of integrating mandatory adaptations to the data systems.
Friedrich Asmus, MD
Chief Medical Officer at Oxular

Wemedoo's technology, expertise, and intimate engagement with our team in the development of the EDC, site/patient interface, and metric capabilities are unique. They provided insight and challenged the underlying assumptions...
Wemedoo's technology, expertise, and intimate engagement with our team in the development of the EDC, site/patient interface, and metric capabilities are unique. They provided insight and challenged the underlying assumptions to ensure the study's goals and objectives were clearly defined and the operating structure matched with the study's intent. Their system is not a one-size-fits-all and they adapt to the needs of the sponsor, sites, physicians, and the study requirements into a system that merges health information/data and can then become not only informative but actionable.
James Kuras
Vice President of Clinical Affairs and Operations at CENTINEL SPINE
Our Proof of Quality
We are ISO 9001 and ISO 27001 certified. Our commitment to robust information security & superior quality management is at the core of our innovative solutions, ensuring reliable & efficient trial management for our clients.


Common Questions
ePRO (Electronic Patient-Reported Outcomes) software allows clinical trial participants to report health outcomes, symptoms, and treatment impacts directly through digital devices. Wemedoo’s oomnia ePRO streamlines this process by capturing cleaner data in real-time, improving patient engagement, and providing clinical sponsors with immediate, integrated insights.
oomnia ePRO uses a responsive web-based interface that works seamlessly on any personal device, including smartphones (iOS and Android), tablets, laptops, and desktop computers. Participants do not need trial-specific "provisioned" devices or app installations. If a patient’s device is lost or replaced, they can resume their assessments instantly via a secure link sent by SMS or email.
Because oomnia ePRO is part of a unified system, data flows directly into the Electronic Data Capture (EDC) system in real time. Questionnaires can be automatically triggered from within a participant’s electronic Case Report Form (eCRF). This eliminates the need for manual data transfers, reducing time-consuming, error-prone, and costly reconciliations.
oomnia ePRO improves data reliability through real-time validation at the point of entry and built-in edit checks, time-stamped responses that prevent backfilling, and automated flagging of inconsistencies, outliers, or missing data. Integrated graphical reports give study staff a real-time overview of instrument completion, allowing clinical teams to follow up with participants immediately to correct discrepancies.
Yes. oomnia ePRO supports fully customizable surveys and questionnaires tailored to any clinical trial protocol. You can configure validated instruments (such as PRO-CTCAE, EQ-5D, and SF-36) or build custom scales from scratch using our intuitive drag-and-drop form designer.
Completing assessments on their own familiar devices is significantly more comfortable for patients than maintaining paper diaries. oomnia ePRO further boosts engagement through multilingual support (allowing patients to use their preferred language) and automated reminders that prompt participants to complete their scheduled assessments on time.
To book a demo, simply click the booking button on our website, select a convenient date in the calendar tool, and fill in your details. We will send you a calendar invitation to meet with an oomnia expert.

Looking to explore more? Get our ePRO flyer for more details.

Read more about the role of ePRO in patient-centric clinical trials.