Real-Time Oversight for Clinical Data Managers and Sponsors
Eliminate manual data reconciliation, resolve discrepancies instantly, and accelerate database lock with an EDC built for complex, global trials.
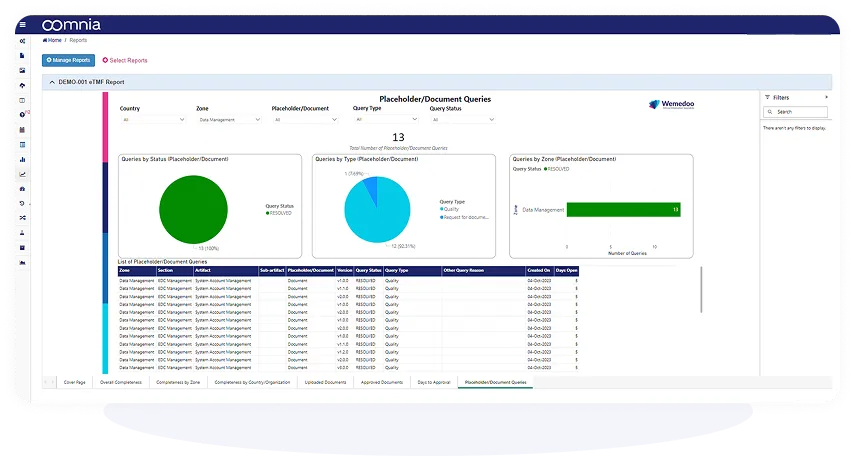
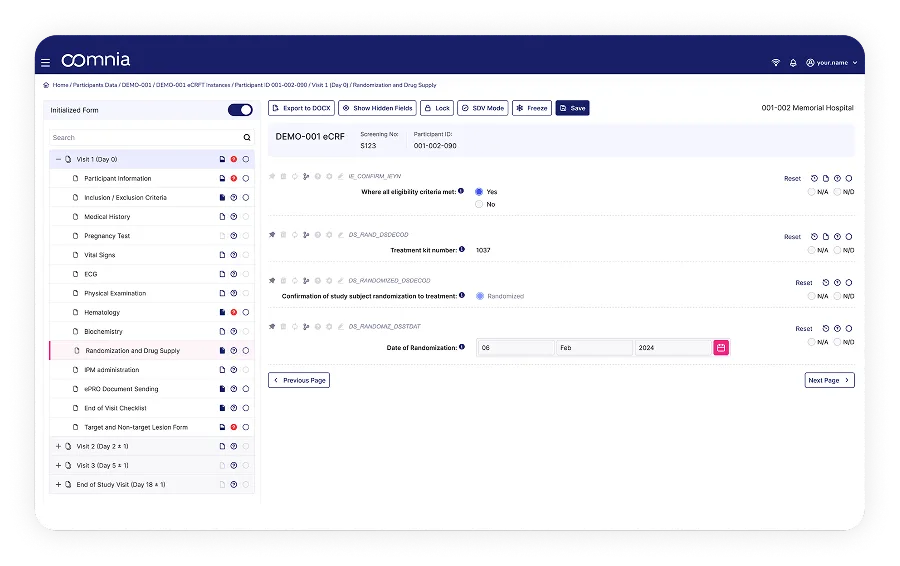
Powerful Dashboards for Real-Time Reporting
Generate custom reports, dashboards and statistical analyses directly within the system. All data is analysis-ready at any time and you decide what and how to present your data.
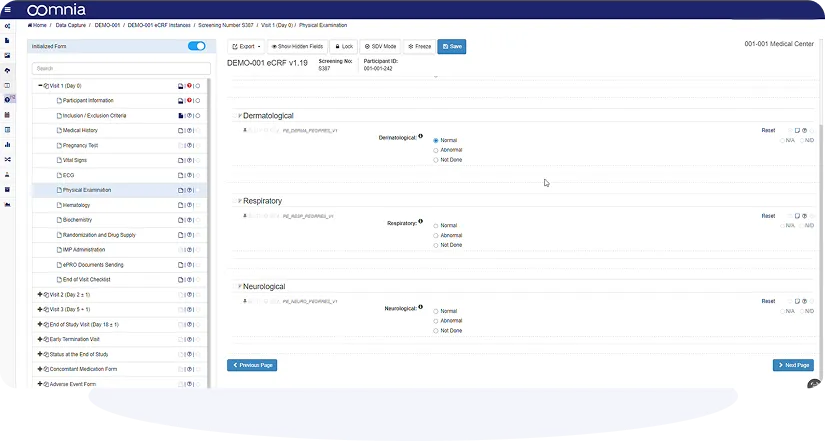
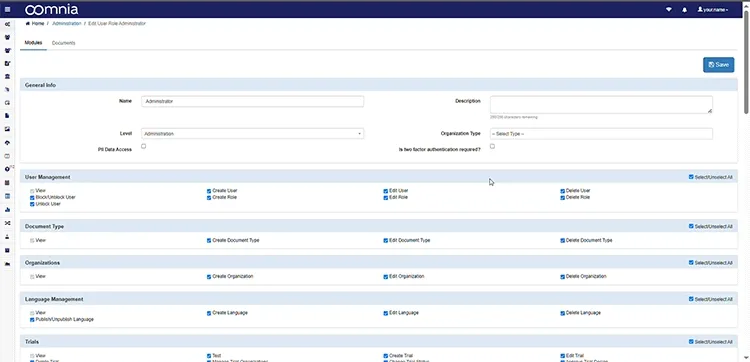
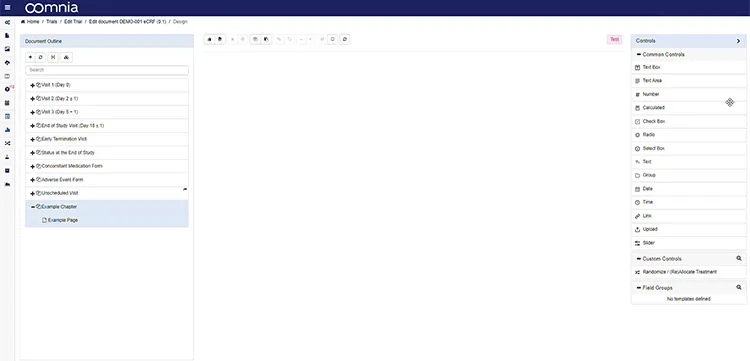
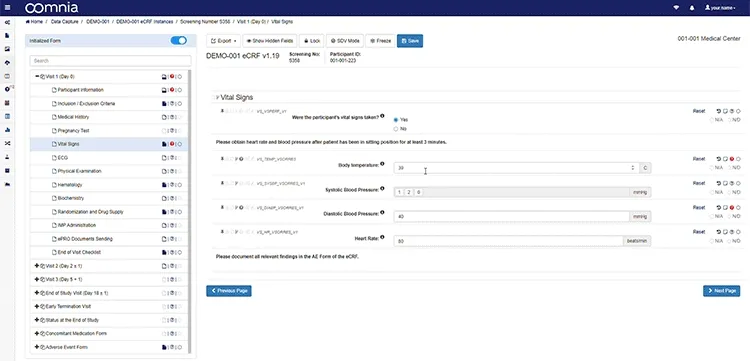
Intuitive User Interface
Positive user experience, intuitive design and powerful no-code user interface allows efficient workflow and productive collaboration.