Article
Why compliance risk increases in modular eClinical setups
Structural compliance risk in modular eClinical architectures
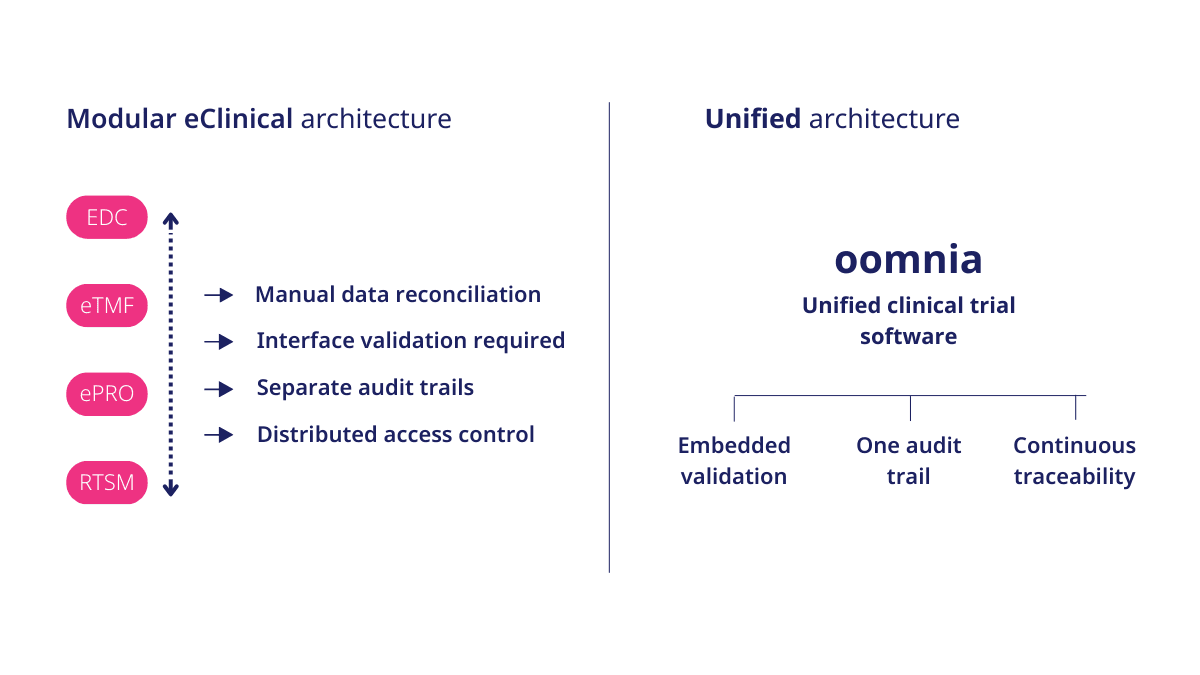
Modular eClinical setups combine specialized technologies such as Electronic Data Capture (EDC), Randomization and Trial Supply Management (RTSM), Electronic Patient‑Reported Outcomes (ePRO), Clinical Trial Management Systems (CTMS), and Electronic Trial Master Files (eTMF) from different vendors.
While this approach allows organizations to select best-of-breed tools, it also introduces architectural fragmentation.
Compliance risk emerges not just because individual systems lack validation, but because fragmented solutions make end‑to‑end control difficult to demonstrate. Even when processes are well designed and each platform is fully validated, structural weaknesses appear at the integration boundaries, where systems exchange data, responsibilities overlap, and traceability becomes reconstructed rather than inherent.
These weaknesses typically manifest as:
- Duplicated validation and oversight evidence
- Reliance on manual reconciliation across systems
- Disconnected audit trails limiting end‑to‑end traceability
- Reactive inspection preparation requiring cross‑vendor coordination
As regulatory expectations evolve toward continuous, demonstrable oversight, these structural gaps are becoming increasingly visible and increasingly consequential during inspections.
These operational challenges are not isolated issues but direct consequences of broader architectural limitations within today’s clinical research technology landscape. As discussed in our analysis “Why eClinical architectures have reached their limits, and a new category is emerging,” fragmented system environments increasingly struggle to provide the level of oversight modern clinical research requires.
Validation multiplicity in a modular eClinical setup and the risk of duplicated evidence
Each system supporting clinical trial activities must undergo documented validation in accordance with applicable regulatory requirements.
In modular environments, validation packages exist per vendor. However, regulatory authorities assess the sponsor’s overall governance framework and its ability to demonstrate control across the entire system landscape.
As system count increases, validation documentation expands:
- Separate user requirement specifications
- Individual installation, operational, and performance qualification documentation
- Independent change management records
- Periodic review reports per platform
- Increasing validation and security documents for each integration point
Validation evidence, access management records, and oversight documentation are often generated separately and later consolidated under inspection pressure.
This duplication increases administrative burden and elevates the risk of inconsistency.
In an interview with Xraised, Dr. Nikola Cihorić reported that one Contract Research Organization (CRO) eliminated more than 1,100 hours of system integration work after replacing a modular technology stack with a unified clinical research information system.
While primarily reflecting operational efficiency, this example illustrates a structural issue common in fragmented system landscapes: integration layers often expand validation scope, increase reconciliation workload, and generate duplicated oversight documentation across systems.
Interface dependencies in modular eClinical architectures and the need for manual reconciliation
Data routinely moves between systems in modular eClinical architectures. Randomization outputs may feed EDC. ePRO data may inform statistical analysis. CTMS exports may support regulatory reporting.
Each interface requires:
- Defined data ownership
- Validated data mapping
- Documented processing specifications
- Reconciliation procedures
- Exception management traceability
In fragmented environments, reconciliation frequently relies on manual cross-system comparison rather than structurally embedded controls.
This reliance on procedural coordination rather than system-level controls increases compliance exposure.
“CSV principles do not fail inside validated systems. However, they could fail in the gaps between them.” — Dusan Goljic, Director of Quality Management
Audit trail fragmentation and data integrity risk in modular eClinical systems
Regulatory frameworks require secure, time-stamped audit trails to ensure clinical data integrity and traceability.
Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have intensified their focus on audit trail compliance during inspections. ¹
In modular setups, audit trails exist in separate applications, so end‑to‑end traceability must be assembled from multiple sources rather than viewed as a single, continuous record.
This fragmentation introduces:
- Gaps in end-to-end data traceability
- Inconsistent timestamp standards
- Distributed role-based access models
- Delayed discrepancy detection
From an inspection standpoint, reconstructing a subject-level data journey across multiple systems becomes procedurally complex.
Vendor oversight and distributed accountability
Good Clinical Practice (GCP) principles require sponsors to maintain oversight of trial-related activities, even when delegated. In modular system environments, multiple providers operate under separate quality management systems and independent release cycles.
Sponsor governance must therefore include:
- Vendor qualification and audit programs
- Ongoing performance monitoring
- Cross-system impact assessment
- Coordinated change management review
- Validation procedures and risk-based assessment to each separate system
When integration failures occur, accountability may appear distributed. Regulatory authorities, however, evaluate the sponsor’s governance model as a unified system.
Distributed accountability increases compliance risk by creating oversight blind spots and complicating inspection defensibility.
Reactive inspection preparation in fragmented eClinical environments
Inspection readiness in modular environments often depends on cross-system evidence consolidation.
When inspectors request end-to-end traceability, from subject randomization through database lock, documentation must be retrieved from multiple platforms.
In fragmented architectures, inspection preparation becomes reactive. Evidence is assembled across vendors rather than derived from structurally embedded traceability.
Inspection defensibility becomes coordination-dependent rather than architecture-driven.
Regulatory evolution: Structural oversight under ICH E6(R3)
The International Council for Harmonisation (ICH) E6 Guideline for Good Clinical Practice (R3) reinforces risk-based quality management and emphasizes proportionate oversight of computerized systems across the clinical trial lifecycle.
Regulatory focus is shifting toward control, traceability, and system accountability, not merely procedural documentation.
As discussed in our analysis of structural oversight under ICH E6(R3) architectural clarity increasingly determines how effectively sponsors demonstrate governance over digital infrastructures.
In modular environments, achieving structural oversight becomes more complex due to distributed validation ownership and fragmented data flows.
Risk profile comparasion
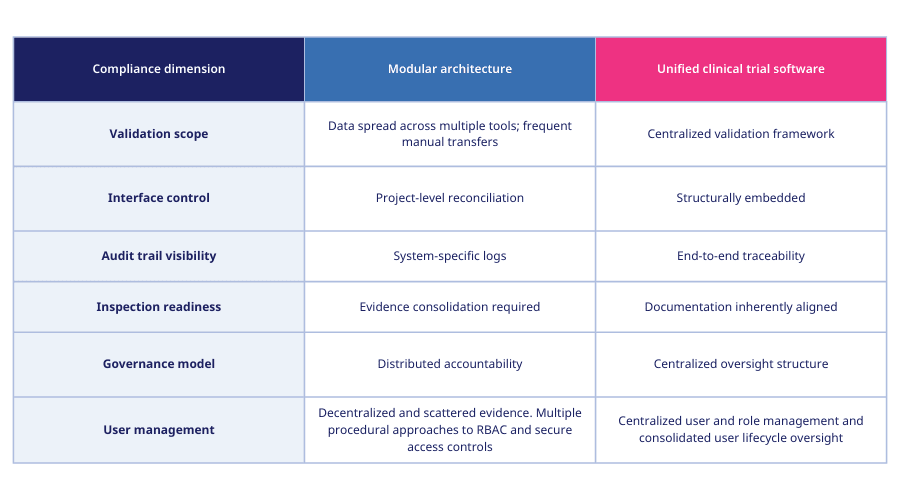
Fragmentation across systems expands the burden of demonstrating compliance control.
How unified clinical trial software reduces structural compliance risk
Compliance risk in modular eClinical setups is not caused by individual system non-compliance. It results from fragmentation.
Unified clinical trial software oomnia reduces:
- Validation duplication
- Manual reconciliation burden
- Audit trail discontinuity
- Oversight diffusion
- Cross‑vendor change‑control complexity
- Evidence fragmentation across systems
- Manual workload at database lock
In oomnia, a fully unified clinical research information system, validation governance and traceability continuity are embedded within the system design, strengthening inspection readiness.
Consclusion
Compliance complexity in modular eClinical setups increases as architectural fragmentation expands.
Regulatory authorities evaluate the sponsor’s ability to demonstrate continuous, end-to-end governance of computerized systems and clinical data. As system fragmentation grows, so does the burden of proof.
In regulated clinical research, structural coherence is not an operational preference. It is a compliance control mechanism.
Discover how oomnia embeds structural compliance control into clinical trial software architecture.
Learn more: https://wemedoo.com/clinical-trial-software-tools/
References:
https://www.clinicalstudies.in/what-regulators-expect-in-an-audit-trail-review/

February 6, 2026
Handling complex data in oncology trials
Blogs

January 28, 2026
Why eClinical architectures have reached their limits, and a new category is emerging
Blogs

January 6, 2026
Clinical trial trends in 2025
Blogs