Article
Handling complex data in oncology trials
For clinical data teams, oncology studies turn routine data work into an ongoing struggle for integrity and completeness. These trials often collect far more patient data than studies in other disease areas, challenging the quality of execution and data oversight. Delays or errors in managing such large and complex datasets can affect patient safety and extend study timelines.
The impact of this data burden is reflected in the fact that oncology trials take nearly 50% longer than trials in other therapeutic areas.¹ Adding to this complexity in execution, patient recruitment is extremely complex and time-consuming, making oncology studies the longest and most challenging in the industry. For data management teams, this means dealing with fragmented information from multiple sources, irregular enrollment patterns, and constantly changing visit schedules.
Without an adequate solution to handle this complexity, timelines stretch, resources strain, and execution slows, impacting the speed at which life-changing therapies reach patients.
Complexity in oncology studies
Oncology trials are among the most demanding in clinical research, requiring the management of extensive and diverse datasets. Frequent protocol amendments, difficult recruitment processes, and elevated dropout rates (often driven by disease progression or adverse events) add further complexity and can lead to significant delays and increased costs.
Tracking cohorts and maintaining data accuracy becomes even more difficult when trials involve multiple sub-groups and cycle-based treatment regimens. For example, a study might test a single drug across patients with the same genetic mutation and different cancer types, or compare several treatments within the same diagnosis. Some early-phase studies even dynamically expand patient groups after initial dose evaluations to accelerate personalized insights. These operational realities frequently push traditional data management systems beyond their limits.
As oncology research evolves toward more adaptive and personalized approaches, technology must evolve as well. Yet many existing solutions were built for an earlier era, defined by static protocols, rigid designs, and single-intervention trials. When these traditional systems reach their limits, they contribute to longer timelines and increased operational risk.
How unified oncology data improves patient safety and trial execution
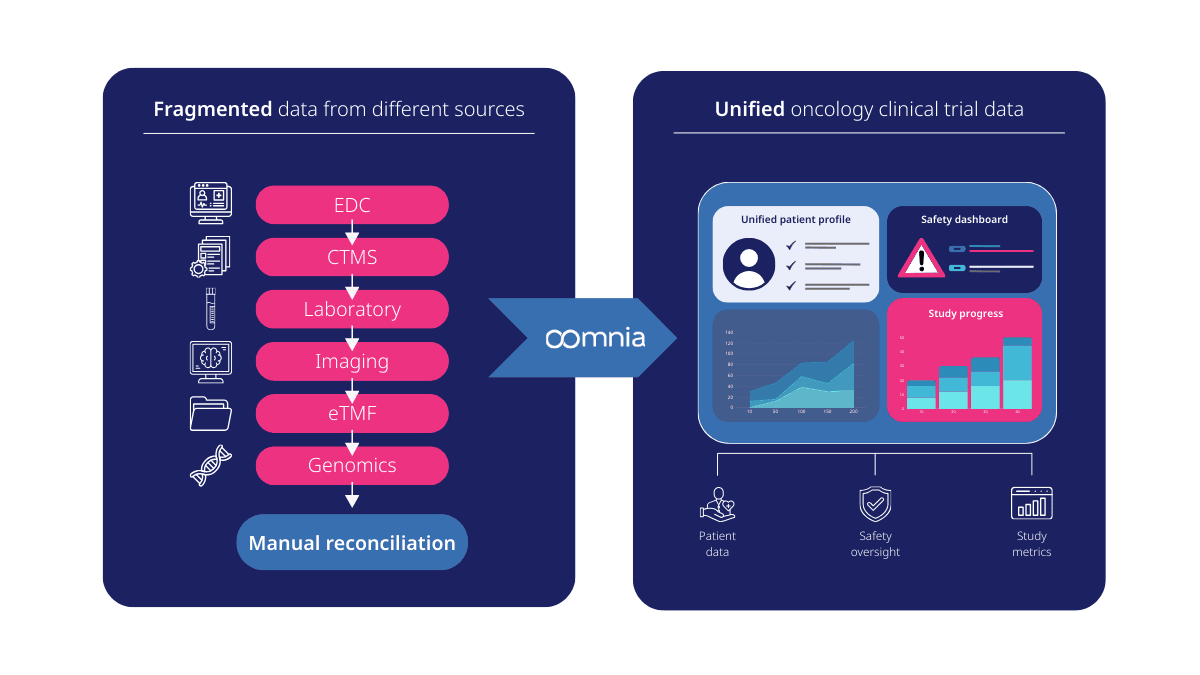
When trial data is scattered across multiple systems, teams often struggle to maintain momentum and oversight. Fragmentation leads to delays, manual reconciliation, and loss of context, increasing the risk of inconsistencies that can affect patient safety and slow decision-making.
Unified data solves this by providing a single, continuously updated view of patient information, overall safety data, and study progress. Integrated patient profiles allow investigators and safety teams to review adverse events and clinical trends in a timely, contextualized manner, supporting more informed safety oversight throughout the trial.
Speaking to the impact of fragmentation on patient safety, Nikola Cihoric, Founder and Chief Product Officer, emphasizes the need for a unified view of clinical data: “Every data point in an oncology trial represents a patient. When information is fragmented, risk increases. When it is unified, safety teams gain the context and visibility they need to identify risk earlier and act with confidence. Oncology trials are complex enough; teams shouldn’t waste time piecing data together to understand what is happening.”
With improved visibility into treatment cycles and visit completion, investigators can better assess protocol adherence, while study coordinators and sponsors can more easily track enrollment status and key milestones. This shared view of clinical and operational data helps reduce errors, supports faster review processes, and strengthens patient protection across complex oncology studies.
oomnia makes this possible by consolidating all oncology trial data into one clinical research information system, delivering real-time visibility and contextualized insights. By connecting essential tools in one place, oomnia supports consistent data review, clearer oversight, and more efficient trial execution.
Big data problem in clinical research
Data management teams play a critical role in clinical research, ensuring that clinical trial data is accurate, consistent, and ready for analysis. However, the rise of big data has transformed this responsibility into a complex challenge, particularly pronounced in oncology.²
Traditional systems and workflows were designed for structured datasets with predictable formats, but modern oncology trials generate massive volumes of diverse data, from electronic health records (EHRs) and lab results to imaging files, omics files, and patient-reported outcomes.
When managed effectively, this data can bring important improvements in clinical research and ensure better treatment results. However, the complexity and scale of these datasets increase the risk of delays, inconsistencies, and data quality issues that need to be addressed to use them effectively.
Growing volume of oncology clinical trial data
The amount of data in oncology is continuously increasing, coming from many different sources like next-generation sequencing (NGS), EHRs, wearable devices, and more.
This includes:
- Clinical data – patient demographics, medical history, treatment details,
- Genomic data – sequencing results, gene expression profiles, mutation analysis,
- Imaging data – MRI, CT, PET scans for tumor detection and monitoring,
- Biomarker data – blood tests, tissues samples, molecular markers,
- Instrument-generated data – outputs from flow cytometry, mass spectrometry, NGS, etc.
- Pharmacological data – drug absorption, metabolism, and effect measurements.
Because of that, the data burden in oncology trials is growing. A single cancer patient’s multiomics profile (combining genomic, transcriptomic, proteomic and other layers), can generate between 2 and 4 terabytes of information, sometimes even more.³
This data should be properly stored, managed, and further analyzed, highlighting the complexity of trial data management and underscoring why oncology research is at the forefront of the clinical big data challenge.
How enrollment issues affect data quality
Enrollment in oncology trials rarely follows a neat, one-time schedule, with participants often joining continuously and across multiple sub-groups.
As patients enter the study at different times, data managers must constantly check which patients qualify for each sub-arm and ensure visit cycles match, otherwise cohort views can become fragmented.⁴
Because of ongoing enrollment, data arrives at different times and in different patterns, making it harder for data management teams to maintain accuracy and completeness. When enrollment is slow or uneven, it affects not only timelines but also the quality and structure of the data received. Combined with the growing volume of data from various sources, these factors contribute to the big data challenge in oncology research.
oomnia addresses these challenges by enabling all patient data to be tracked in one place and updated easily, ensuring accuracy, completeness, and efficiency throughout the complex trial process.
Big data and Artificial Intelligence (AI)
As we discussed in our recent blog, AI plays an important role in advancing clinical research, being one of the tools helping with big data problems in oncology trials.
AI is changing how this data is processed and interpreted, enabling faster and more accurate insights. For example, NGS produces high-throughput genomic data that helps identify biomarkers and guide targeted therapies, while AI-driven algorithms can be employed to gain relevant clinical information that will decrease errors.⁵ Beyond genomics, AI also reduces manual workloads, standardizes analysis, and improves diagnostic accuracy in medical imaging and digital pathology.
In complex oncology trials, instead of relying on manual document sorting and time-consuming eligibility checks, CROs now use AI to automate these processes. Advanced models analyze genetic profiles and EHRs to quickly identify suitable participants, making recruitment faster and more precise.
Beyond patient recruitment, AI also organizes trial documents in real time and flags inconsistencies, reducing administrative burden and improving data accuracy. This integrated approach helps teams manage complexity of the trials more efficiently and keep studies on track.
Turning complexity into clarity
Timelines in oncology trials matter because delays can slow the development of therapies that patients need. The struggle lays in how many parts need to be coordinated, and much of the friction comes from fragmentation.
When teams use separate tools for document management, cohort logs, screening and enrollment, adverse event tracking, and analytics, even small misalignments can lead to big delays.
A practical way to reduce that complexity is to consolidate workflows into a unified solution, where the same system supports each step of the trial. In such a setup, each data element is traceable end-to-end in real time, and updates are seen consistently.
oomnia exemplifies this approach by enabling end-to-end traceability of clinical, safety, and operational data in real time. That helps teams track all the trial activities in one place and reduce redundant data entry.
Most important part of turning complexity into clarity is giving investigators, sponsors, and CROs a right solution which is going to simplify the trial execution. Also, in oncology research, execution quality directly influences patient safety and reliability of study outcomes.
By standardizing how data is captured, linked, and reviewed in oncology studies, focus is kept on where it belongs - generating high-quality research that brings needed therapies to patients sooner.
References
CenterWatch. (2025, October 29). Emerging challenges in oncology trials: Enrollment, protocol deviations, and growing data. CenterWatch Insights. Accessed January 8, 2026. https://www.centerwatch.com/insights/emerging-challenges-in-oncology-trials/
Hesse, Walter. “Clinical Data Management in the Era of Big Data: Challenges and Opportunities.” Pharmacoeconomics 8 (2023): 196.
Barker, A. D., & Lee, J. S. H. (2022). Translating “Big Data” in oncology for clinical benefit: Progress or paralysis. Cancer Research, 82(11), 2072–2075. https://doi.org/10.1158/0008-5472.CAN-22-0100
Badhrinarayanan, S. (2025, October 14). The challenge of patient enrollment in clinical trials. NCODA. Accessed January 9, 2026. https://www.ncoda.org/news/the-challenge-of-patient-enrollment-in-clinical-trials/
Dlamini, Z., Francies, F. Z., Hull, R., & Marima, R. (2020). Artificial intelligence (AI) and big data in cancer and precision oncology. Computational and Structural Biotechnology Journal, 18, 2300–2311. https://doi.org/10.1016/j.csbj.2020.08.019

March 11, 2026
Why compliance risk increases in modular eClinical setups
Blogs

January 28, 2026
Why eClinical architectures have reached their limits, and a new category is emerging
Blogs

January 6, 2026
Clinical trial trends in 2025
Blogs